BENICCI – VOLUMETRIC SPIROMETER (Incentive spirometer)
Instructions for Use (IFU)
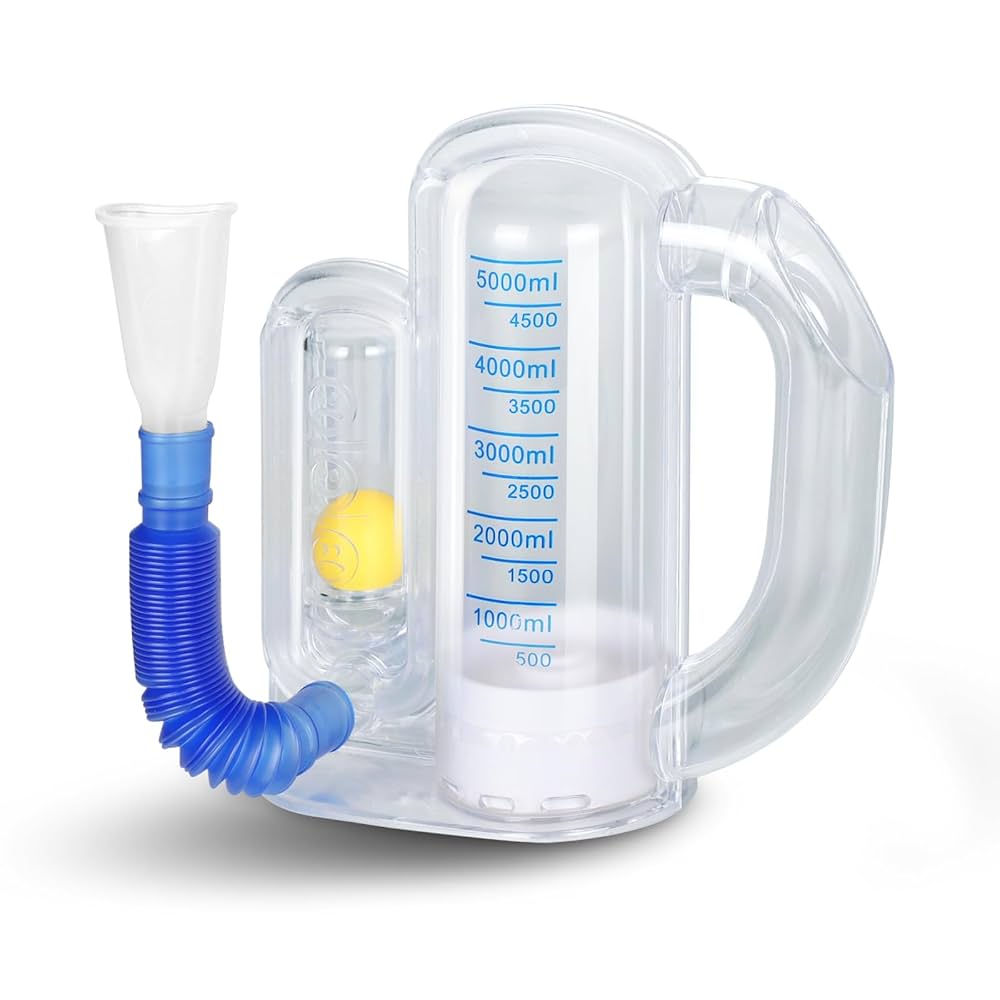
Volumetric Incentive Spirometer
Device Identification
Device Name: Volumetric Incentive Spirometer
Intended Purpose: A non-invasive device designed to encourage voluntary deep breathing to improve lung function, promote lung expansion, and prevent postoperative pulmonary complications (e.g., atelectasis).
Indications for Use
- To promote sustained maximal inspiration.
- To aid in lung expansion following thoracic or abdominal surgery.
- To prevent or treat pulmonary complications such as atelectasis.
- For patients in hospitals or home care who require respiratory rehabilitation.
Contraindications
- Patients unable to follow instructions or cooperate with the therapy.
- Patients with unstable cardiovascular status (e.g., recent myocardial infarction, severe arrhythmia).
- Patients with untreated pneumothorax.
- Use with caution in patients with severe respiratory distress unless under medical supervision.
Warnings
- Device should be used only under the direction of a healthcare professional.
- Not for continuous inhalation therapy—intended for intermittent deep breathing exercises.
- Ensure the device is clean and dry before each use.
- Do not share between patients unless reprocessed according to hospital infection-control protocols.
- Replace immediately if damaged or if any parts are missing.
Precautions
- Monitor patients at risk of dizziness, light-headedness, or hyperventilation.
- Use in a seated or semi-upright position to avoid aspiration risk.
- Children should use the device only under adult supervision.
- Clean device regularly as per instructions to prevent infection.
Instructions for Use
- Sit upright or in a comfortable semi-upright position.
- Hold the spirometer in an upright position.
- Place the mouthpiece in your mouth and seal your lips tightly around it.
- Exhale normally.
- Inhale slowly and deeply through the mouthpiece, raising the piston/ball(s) inside the chamber to the prescribed target volume.
- Hold your breath for at least 3–5 seconds at maximum inspiration, then exhale slowly.
- Remove the mouthpiece and relax before repeating.
- Repeat the exercise 10–15 times per session, or as instructed by your healthcare provider.
- Perform exercises every 1–2 hours while awake, unless otherwise directed.
Cleaning and Maintenance
- After each session, remove the mouthpiece and wash with warm water and mild detergent.
- Rinse thoroughly and allow to air dry completely before reassembly.
- Do not expose to excessive heat or direct sunlight.
- Replace device if cracks, discoloration, or damage appear.
Storage Conditions
- Store in a clean, dry place at room temperature.
- Keep away from dust, moisture, and direct sunlight.
Disposal
- Dispose of the device according to hospital waste protocols or local regulations for plastic medical devices