TT – GUARD

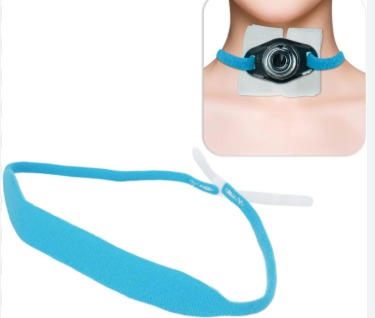
Tracheostomy Tube Holder
Device Identification
* Device Name: Tracheostomy Tube Holder
* Intended Use: To secure a tracheostomy tube in place and help prevent accidental decannulation.
* Device Classification:
* EU MDR (2017/745): Class I, non-invasive, non-sterile, single-use medical device.
* US FDA: Class I, 21 CFR 880.5780, Tracheostomy Tube and Tube Holder (exempt).
Indications for Use
The tracheostomy tube holder is indicated for securing tracheostomy tubes in adult and pediatric patients in hospital, long-term care, and home care environments.
Contraindications
* Patients with known hypersensitivity to materials used in the device.
* Patients with skin breakdown, open wounds, or infection at the neck area.
4. Warnings
* Single-patient, single-use only. Do not reuse. Reuse may lead to cross-contamination, infection, or device failure.
* Do not use if the package is open or damaged.
* Ensure correct sizing before use.
* The holder must not be applied too tightly—allow space for at least one finger between the holder and the patient’s neck.
* Frequent monitoring is required to avoid pressure injury, skin breakdown, or airway compromise.
* Do not modify the device.
Precautions
* Replace the holder immediately if it becomes wet, soiled, or damaged.
* Change at least every 24–72 hours, depending on institutional policy and patient condition.
* Inspect skin condition regularly and reposition holder if necessary to reduce localized pressure.
Instructions for Use
1. Perform hand hygiene and wear gloves.
2. Position the patient comfortably and stabilize the tracheostomy tube.
3. Remove the old holder carefully while maintaining tube security.
4. Slide the new holder under the patient’s neck with the soft side facing the skin.
5. Secure each end of the holder to the tracheostomy tube flanges.
6. Adjust the hook-and-loop fastening (Velcro) to achieve a snug but comfortable fit.
7. Verify that the tube is midline and stable.
8. Confirm one finger can fit between the holder and the neck.
9. Dispose of the used holder in accordance with hospital infection control protocols.
Storage Conditions
* Store in a clean, dry place at room temperature.
* Protect from direct sunlight, excessive heat, and moisture.
Shelf Life
Use before the expiry date printed on the packaging.
Disposal
Dispose of the used holder in accordance with hospital/clinical waste management policies for medical devices.